How the MAGNET study delivered a 0% false negative rate (FNR) for patients post-NAT
Since the landmark ACOSOG Z1071 trial, leading physicians have pondered how to improve the accuracy of axillary assessment after neoadjuvant chemotherapy. A new Spanish study has now validated the ideal approach for TAD…

According to the MAGNET study, published in July 2022, the solution to achieving optimal results is to retrieve the clipped node using a Magseed® marker, alongside sentinel lymph node biopsy (SLNB). This procedure is otherwise known as Targeted Axillary Dissection (TAD), which originated at the University of Texas MD Anderson Cancer Center.
The multicenter study assessed a total of 81 patients with known-positive lymph nodes, marked with a Magseed® either before or after neoadjuvant therapy (NAT).
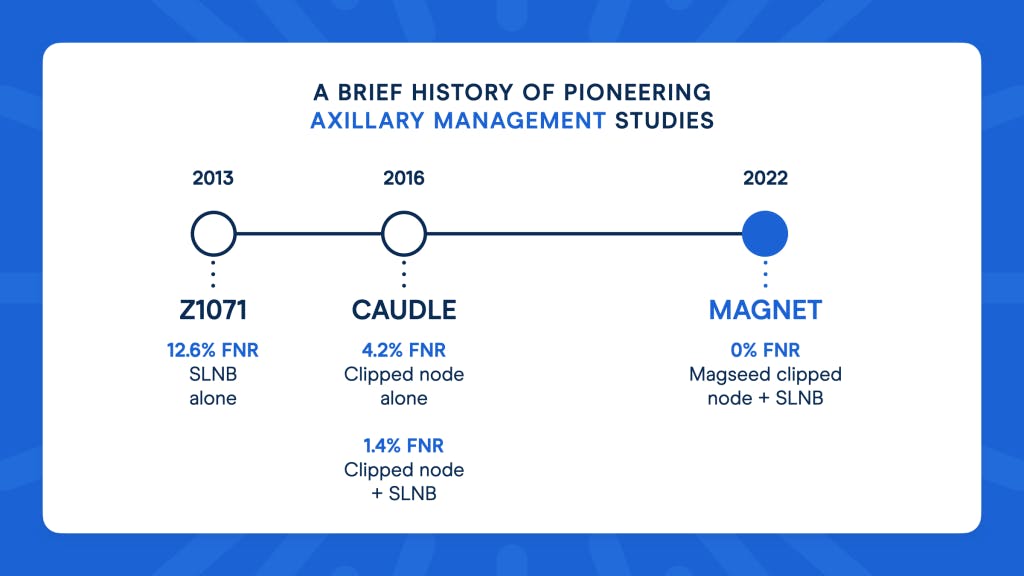
Of those 81 patients, all seeds were successfully retrieved, with 34 patients demonstrating a pathologic complete response (pCR). Importantly, the study also showed a false negative rate (FNR) of 0%.
As the paper acknowledged, these findings could represent a significant breakthrough in reducing the necessity for aggressive axillary lymph node dissection (ALND) surgery.
How Z1071 opened up the conversation
To fully understand the magnitude of the MAGNET trial’s results, you have to first go back almost a decade to the release of the ACOSOG Z1071 paper.
In this groundbreaking paper, a team led by Judy Boughey, M.D. of the Mayo Clinic set out to establish whether SLNB provided enough accuracy for assessing nodal response post-NAT. This was driven by a desire to reduce the number of ALND surgeries being performed unnecessarily, and reduce long-term patient side-effects - such as lymphedema.
To validate their findings, they captured and reported on 756 patients across 136 institutions. At first glance, the results were disappointing.
They found that in patients with SLNB alone post-NAT, the FNR worked out as 12.6%, significantly higher than the acceptable rate they had previously set of 10%.

However, something important was observed in the Z1071 paper findings. In 170 patients, a biopsy-proven positive node was clipped before NAC. They found that in 24% of these patients, the SLNB missed these clipped nodes, thus driving down the accuracy of the findings.
When the clipped node was removed as part of the SLNB, the FNR dropped to just 6.8% - a much more acceptable result.
The emergence of TAD as an advanced approach
At the University of Texas MD Anderson Cancer Center, they’d been watching these results and were encouraged by the outcomes from the clipped nodes.
In 2016, they released a paper (Caudle, 2016), which looked at reducing the FNR further - firstly by performing the clipped node approach alone on a subsection of patients, and then performing the full TAD procedure on another group. The findings were impressive.

For clipped node alone, a lower 4.2% FNR was reported - but for the clipped node and SLNB together, this dropped even further to just 1.4%.
A new gold standard for axillary surgery post-NAT had emerged and was soon being used routinely around the world - including by the MAGNET study authors.
Utilising Magseed for TAD in the MAGNET study
As routine Magseed® users, the authors of the MAGNET study decided that they wanted to see the results for themselves, with their preferred localization marker.
They marked the nodes both pre-NAT (44 patients) and post-NAT (37 patients), monitoring nodal response by ultrasound.
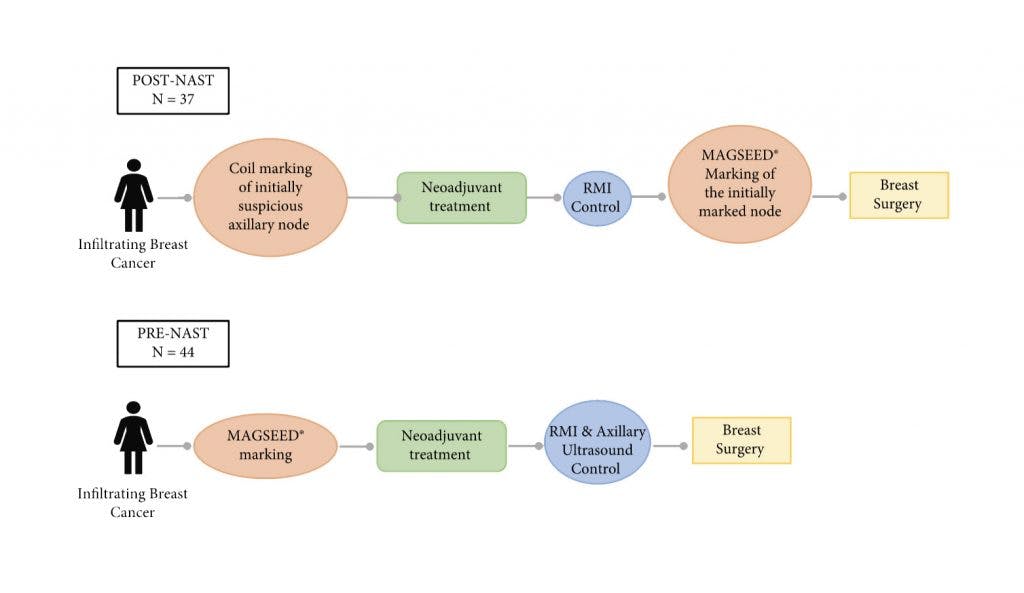
Aside from achieving a 100% retrieval rate and 0% FNR across all patients studied, they saw many other encouraging results:
- All Magseed® marked nodes were successfully retrieved in just over 10 minutes, guided by the Sentimag®.
- The concordance rate between Magseed® marked nodes and sentinel lymph nodes was 81.5% - similar to many other publications, including Simons et al (2021).
- Discordant cases were due to positive Magseed® marked nodes and negative sentinel nodes, suggesting Magseed® could be a more sensitive method for detecting metastasis.
- No significant differences were found between Post-NAT (37) and Pre-NAT (44) placements - Magseed® was equally reliable in both settings.
With these findings and numerous other Magseed® for TAD studies, it is becoming clearer that it is able to deliver similar results to radioseeds, but without the burden and expense of setting up a radioactive seed program.
Speaking about the results, the study author Dr Maria Martínez told us, "The Magseed magnetic marker, in working groups with extensive experience in axillary ultrasound staging in breast cancer and on a strict selection of patients with cN1 axilla (1-3 suspected nodes), has proven to be a safe alternative for node marking in patients with positive axilla and indication for neoadjuvant therapy."
"In centres without nuclear medicine department, Magseed allows adequate control of the FN rate and guarantees the recovery of the initially marked node in all cases."Dr Maria Martínez
Validating the findings in future studies
As with the ACOSOG Z1071 study, the researchers have emphasised the need to continue validating these findings further to avoid misdiagnosis.
This process has already begun, with over 1,000 Magseed® TAD patients featured in clinical data, and plenty more studies in the pipeline. One of these is the MAGELLAN trial, again led by the University of Texas MD Anderson Cancer Center.
MAGELLAN will be using Magseed® as their sole node marker, and the researchers will be placing the seed at the time of biopsy. This will then be followed up by ultrasound monitoring and surgery to remove the marked node, in order to measure the effectiveness of this method.
“There is potential for magnetic seeds to be the definitive axillary node marker”Abigail Caudle, M.D. - University of Texas MD Anderson Cancer Center
For now though, the MAGNET study has set an impressive benchmark for others to build on. With every passing study, it’s becoming apparent that the future of axillary management is Magseed®.
→ Find out more about TAD from Abigail Caudle, M.D.